ETP in pharmaceutical industry addresses one of the most complex environmental challenges manufacturing faces. Pharmaceutical wastewater carries high Chemical Oxygen Demand (COD) levels exceeding 5,000-10,000 mg/L, along with dangerous heavy metals like lead, mercury and cadmium. These effluents contain toxic chemicals and active pharmaceutical ingredients with variable pollutant loads that require specialized treatment before environmental discharge.
We have created this detailed guide to help you understand how effluent treatment plant in pharmaceutical industry works. You will learn the complete working principles of Pharma ETP and detailed treatment stages. You will also learn how to interpret an ETP plant diagram. This piece breaks down each treatment phase to optimize your facility’s wastewater management and regulatory compliance.
What is ETP in Pharmaceutical Industry and Why It Matters

An Effluent Treatment Plant treats wastewater generated from pharmaceutical manufacturing units before discharge into the environment or reuse within facilities. Pharma ETPs handle uniquely complex effluents containing solvents, active pharmaceutical ingredients, chemicals used in synthesis and cleaning agents, unlike general industrial wastewater systems. These systems employ physical, chemical and biological processes to remove pollutants and purify wastewater to meet environmental regulations.
ETP Full Form in Pharmaceutical Industry
ETP stands for Effluent Treatment Plant. These installations purify water by eliminating chemicals and hazardous materials in pharmaceutical manufacturing, allowing treated water to be recycled or released with minimal environmental effect. The main goal centers on removing toxic, hazardous and non-biodegradable substances from wastewater while ensuring treated effluents comply with local environmental discharge standards.
Key Differences Between Pharma ETP and Industry ETP
Pharmaceutical wastewater is different by a lot from other industrial effluents due to the presence of organic and inorganic materials, solvents and active pharmaceutical ingredients. The removal of APIs poses a major concern owing to possible risks they present to the environment and human health.
Pharma ETPs rely on biological treatment techniques such as activated sludge processes and bioreactors to break down APIs and other organic chemicals. Advanced oxidation methods provide additional treatment for recalcitrant compounds. The pharmaceutical sector focuses on wastewater reuse, with facilities recovering and recycling APIs to minimize environmental effect. Specialized analytical techniques including High-Performance Liquid Chromatography (HPLC) and mass spectrometry guarantee efficient clearance of APIs and medicinal chemicals through monitoring.
Regulatory Requirements for Pharmaceutical Effluent Discharge
Pharmaceutical companies must comply with stringent regulations regarding wastewater discharge. The EPA promulgated the Pharmaceutical Manufacturing Effluent Guidelines and Standards (40 CFR Part 439) in 1976 and amended the regulation in 1983, 1998 and 2003. These guidelines apply to facilities divided into five subcategories: Fermentation Products, Extraction Products, Chemical Synthesis Products, Mixing/Compounding and Formulation, and Research.
National Pollution Control Boards in different countries and European Union Directives on Wastewater Discharge provide additional regulatory oversight. The Central Pollution Control Board dictates national discharge levels in effluent in India, while State Pollution Control Boards enforce environment-related regulations at the state level. Non-compliance results in heavy penalties, operational shutdowns or legal action.
Characteristics of Pharmaceutical Wastewater
Pharmaceutical manufacturing generates wastewater streams with distinct pollution profiles that separate them from conventional industrial effluents. Understanding these characteristics helps facilities design appropriate treatment systems to remove contaminants.
High Chemical Oxygen Demand (COD) and BOD Levels
Pharmaceutical wastewater exhibits very high organic pollution indicators. COD concentrations range from 1,000 to 10,000 mg/L across manufacturing processes. Research and development facilities record combined waste values reaching 5,716 mg/L. BOD levels span 500-3,000 mg/L during production operations, and R&D units generate effluents containing 1,385 mg/L. These high values stem from refractory organics and toxic pollutants that resist conventional biological degradation. The pollutant loads show negligible BOD in many cases. COD remains substantially higher, showing low biodegradability due to concentrated refractory organic content.
Presence of Active Pharmaceutical Ingredients (APIs)
APIs appear throughout environmental matrices in water, soil, sediment and sludge. Surface waters near drug manufacturing hubs contain APIs at elevated concentrations. Ranges fall below 100 ng/L in surface and groundwater and below 50 ng/L in treated drinking water. Direct releases from pharmaceutical manufacturing contribute 2 percent of total environmental pharmaceutical loads but create high-concentration hotspots in local waterways. These bioactive compounds maintain biological activity after environmental release. They cause harmful effects on aquatic ecosystems and contribute to antimicrobial resistance.
Heavy Metals and Suspended Solids Content
Pharmaceutical effluents contain heavy metals following the concentration trend: Fe >> Zn > Ni > Pb > Mn > Cr6+ > Cd > Cu. Iron concentrations reach 10.86 mg/L, and this is a big deal as it means permissible limits are exceeded. Suspended solids range from 100-2,000 mg/L in manufacturing processes.
pH Fluctuations and Toxic Compounds
Wastewater pH varies from 1 to 11 due to residual acids and bases from synthesis processes. Measured ranges span 3.03 to 10.08. This acidity conditions the toxicity of chemical derivatives, and substances show over 1,000-fold toxicity changes between pH 5 and pH 9.
Working Principle of ETP: Complete Treatment Process
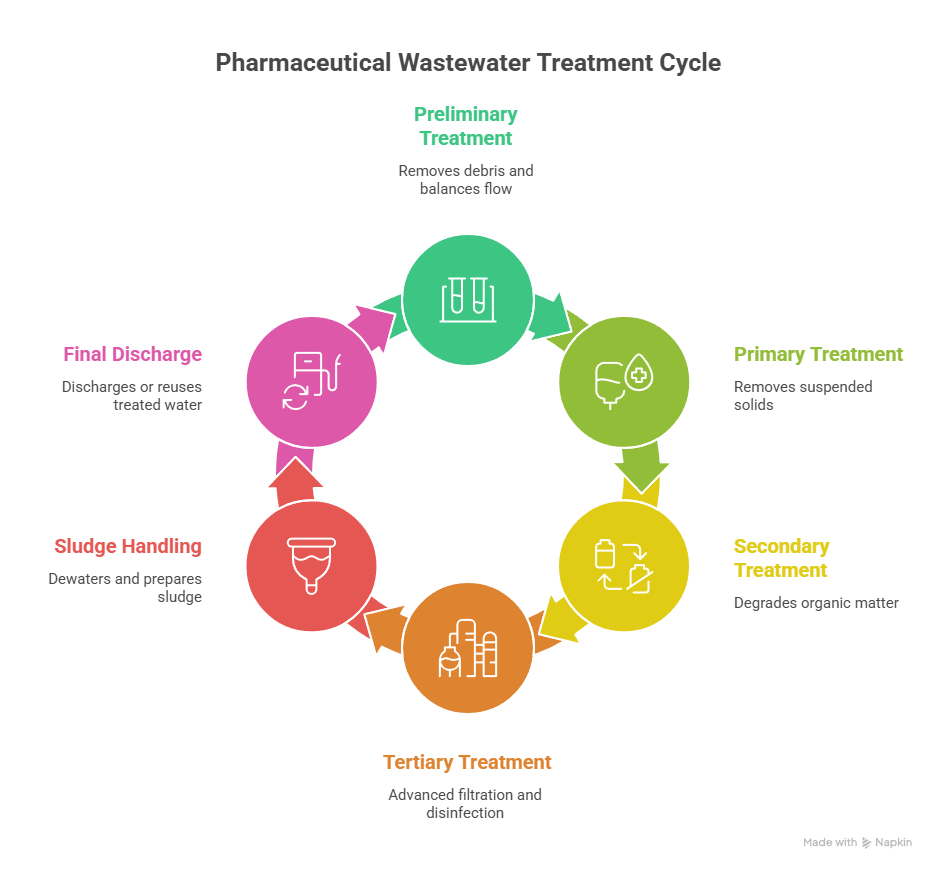
Treatment operations in pharmaceutical facilities follow a multi-stage integrated approach that manages variability, toxicity and stringent regulatory limits through sequential purification phases.
Stage 1: Preliminary Treatment (Screening and Equalization)
Raw effluent enters mechanical screening systems that remove large debris, plastics and solid materials. This prevents downstream equipment damage. Wastewater flows into equalization tanks where it stays for 8 to 12 hours. The tanks balance hydraulic loads and maintain consistent flow rates by mixing and homogenizing wastewater streams. Floating aerators provide continuous agitation to achieve homogeneous effluent before it advances to subsequent stages. pH levels receive monitoring and adjustment at this phase. Wastewater pH should fall between 5.5 and 9.0.
Stage 2: Primary Treatment (Sedimentation and Chemical Coagulation)
Chemical coagulation using alum, PAC or polymers follows preliminary treatment. Dissolved Air Flotation or tube settlers remove flocculated solids and reduce TSS to below 100 mg/L. Organic load drops by a lot before biological treatment. This stage removes 50-60% of suspended solids.
Stage 3: Secondary Biological Treatment (Activated Sludge and MBBR)
MBBR or Activated Sludge Process makes aerobic degradation possible and achieves 70-80% BOD removal. This combined aerobic strategy keeps BOD levels below 30 mg/L, even in toxic and variable wastewater conditions. MBBR systems employ plastic media with high surface areas for biofilm development. Sludge recycling becomes unnecessary.
Stage 4: Tertiary Treatment (Advanced Filtration and Disinfection)
Membrane Bioreactors provide high biomass retention. Ultrafiltration and Reverse Osmosis achieve >99% TDS rejection. Advanced Oxidation Processes such as ozone, UV and Fenton’s reagent destroy refractory pollutants. These systems make up to 95% water recovery possible.
Stage 5: Sludge Handling and Dewatering
Mechanical sludge dewatering systems reduce sludge moisture to 20-30%. Centrifuges or filter presses make safe handling and disposal possible.
Stage 6: Final Discharge or Water Reuse
Treated effluent that meets regulatory standards gets discharged or reused for cooling, cleaning and vehicle washing.
ETP Plant Diagram: Understanding the Process Flow
Process flow diagrams provide visual representations of sequential treatment operations in pharmaceutical ETP systems. Operators can identify equipment locations, monitor treatment efficiency and troubleshoot operational issues when they understand these diagrams.
Key Components in Pharma ETP Plant Diagram
A typical pharma ETP plant diagram has screens, sedimentation tanks, aeration tanks, clarifiers, tertiary treatment units and filtration systems. The effluent collection sump holds 50 KL capacity and feeds into bar screen chambers. Equalization tanks maintain 45 KL capacity each. Primary settling tanks with tube settlers come next. SAFF reactor tanks provide 60 KL capacity for biological treatment, while secondary settling tanks handle biomass separation. Pressure sand filters and activated carbon filters complete the treatment train before final discharge.
Flow Sequence from Inlet to Discharge
Wastewater enters through collection systems and undergoes screening to remove debris. It then flows into equalization tanks for homogenization. pH correction occurs before coagulation and flocculation processes. The flow continues through sedimentation and biological treatment stages. Tertiary treatment with filtration and disinfection follows. Treated effluent either discharges into the environment or gets reused for various purposes.
Critical Control Points in Treatment Flow
Decision-trees can determine critical control points in HACCP systems. This makes process monitoring more logical. Flow diagrams require on-site verification during all operational stages and hours.
Conclusion
We covered everything in ETP systems for pharmaceutical manufacturing, from preliminary screening through advanced tertiary treatment. You now understand how these specialized plants handle complex wastewater containing APIs, heavy metals and toxic compounds through sequential purification stages. You can better optimize your facility’s effluent management, ensure regulatory compliance and contribute to environmental protection. Becoming skilled at these treatment principles positions your operations for eco-friendly pharmaceutical manufacturing, in fact.
Key Takeaways
Understanding ETP systems in pharmaceutical manufacturing is crucial for environmental compliance and sustainable operations. Here are the essential insights every pharmaceutical professional should know:
- Pharmaceutical wastewater contains extremely high COD levels (5,000-10,000 mg/L) and toxic APIs requiring specialized multi-stage treatment beyond standard industrial systems.
- ETP treatment follows six sequential stages: preliminary screening, chemical coagulation, biological treatment, tertiary filtration, sludge dewatering, and final discharge or reuse.
- Regulatory compliance demands meeting stringent EPA guidelines (40 CFR Part 439) with specialized monitoring using HPLC and mass spectrometry for API removal verification.
- Advanced treatment technologies like MBBR, membrane bioreactors, and advanced oxidation processes achieve 95% water recovery while removing refractory pharmaceutical compounds.
- Critical control points throughout the treatment flow require continuous monitoring of pH (5.5-9.0), BOD reduction (70-80%), and heavy metal removal to prevent environmental contamination.
Effective pharmaceutical ETP management combines technical expertise with regulatory knowledge to transform hazardous wastewater into environmentally safe effluent, supporting both operational efficiency and environmental stewardship in modern pharmaceutical manufacturing.
Frequently Asked Questions
Q1. What is the basic purpose of an Effluent Treatment Plant in pharmaceutical manufacturing?
An Effluent Treatment Plant (ETP) in pharmaceutical manufacturing treats wastewater generated during production processes before environmental discharge or reuse. It employs a combination of physical, chemical, and biological processes to remove pollutants, toxic chemicals, active pharmaceutical ingredients, and hazardous materials, ensuring the treated water meets regulatory standards and can be safely released or recycled within facilities.
Q2. What makes pharmaceutical wastewater different from other industrial effluents?
Pharmaceutical wastewater contains uniquely complex pollutants including active pharmaceutical ingredients (APIs), solvents, synthesis chemicals, and cleaning agents. It exhibits extremely high Chemical Oxygen Demand (COD) levels ranging from 5,000-10,000 mg/L, dangerous heavy metals like lead and mercury, and toxic compounds that resist conventional biological degradation. These characteristics require specialized treatment systems beyond standard industrial wastewater facilities.
Q3. What are the main stages involved in pharmaceutical wastewater treatment?
Pharmaceutical ETP systems operate through six sequential stages: preliminary treatment (screening and equalization), primary treatment (sedimentation and chemical coagulation), secondary biological treatment (activated sludge and MBBR), tertiary treatment (advanced filtration and disinfection), sludge handling and dewatering, and final discharge or water reuse. Each stage targets specific contaminants to progressively purify the wastewater.
Q4. What regulatory standards must pharmaceutical companies meet for effluent discharge?
Pharmaceutical facilities must comply with EPA’s Pharmaceutical Manufacturing Effluent Guidelines and Standards (40 CFR Part 439), along with National Pollution Control Board requirements and local environmental regulations. These standards mandate specific limits for pollutants, require specialized monitoring using techniques like HPLC and mass spectrometry, and enforce strict penalties including operational shutdowns for non-compliance.
Q5. How effective are modern ETP systems in treating pharmaceutical wastewater?
Modern pharmaceutical ETP systems achieve 70-80% BOD removal through biological treatment and can recover up to 95% of water through advanced technologies like membrane bioreactors and reverse osmosis. These systems successfully reduce suspended solids to below 100 mg/L, maintain pH levels between 5.5-9.0, and remove over 99% of total dissolved solids, enabling safe environmental discharge or facility reuse.






